- Konen, J.M.; Fradette, J.J.; Gibbons, D.L. The Good, the Bad and the Unknown of CD38 in the Metabolic Microenvironment and Immune Cell Functionality of Solid Tumors. Cells 2020, 9, 52. https://doi.org/10.3390/cells9010052
- Camacho-Pereira J, Tarragó MG, Chini CCS, Nin V, Escande C, Warner GM, Puranik AS, Schoon RA, Reid JM, Galina A, Chini EN. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metab. 2016 Jun 14;23(6):1127-1139. doi: 10.1016/j.cmet.2016.05.006. PMID: 27304511; PMCID: PMC4911708.
- Liu L, Su X, Quinn WJ 3rd, Hui S, Krukenberg K, Frederick DW, Redpath P, Zhan L, Chellappa K, White E, Migaud M, Mitchison TJ, Baur JA, Rabinowitz JD. Quantitative Analysis of NAD Synthesis-Breakdown Fluxes. Cell Metab. 2018 May 1;27(5):1067-1080.e5. doi: 10.1016/j.cmet.2018.03.018. PMID: 29685734; PMCID: PMC5932087.
- Yang Y, Sauve AA. NAD(+) metabolism: Bioenergetics, signaling and manipulation for therapy. Biochim Biophys Acta. 2016 Dec;1864(12):1787-1800. doi: 10.1016/j.bbapap.2016.06.014. Epub 2016 Jun 29. PMID: 27374990; PMCID: PMC5521000.
- Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nat Rev Mol Cell Biol. 2021 Feb;22(2):119-141. doi: 10.1038/s41580-020-00313-x. Epub 2020 Dec 22. PMID: 33353981; PMCID: PMC7963035.
- Stein LR, Imai S. The dynamic regulation of NAD metabolism in mitochondria. Trends Endocrinol Metab. 2012 Sep;23(9):420-8. doi: 10.1016/j.tem.2012.06.005. Epub 2012 Jul 21. PMID: 22819213; PMCID: PMC3683958.
NAD+ is a life-preserving coenzyme. Without NAD+, metabolic homeostasis collapses, and cells become vulnerable to stress-related damage. Thus, identifying new modalities to sustain NAD+ levels is critical for healthy aging.
In the search for effective aging interventions, scientists from leading research institutions have honed in on nicotinamide adenine dinucleotide (NAD+), a vital molecule found in all living cells. NAD+ is a life-preserving coenzyme – a helper molecule that allows enzymes to catalyze reactions necessary for many cellular functions. NAD+ plays a key role in regulating mitochondrial functions and energy metabolism. However, cellular NAD+ levels gradually decline with age, and without a sufficient NAD+ pool, metabolic homeostasis collapses. Furthermore, in the absence of sufficient NAD+, our genetic blueprints (DNA) become vulnerable to unwarranted changes that animal studies have shown to promote several age-related health conditions, such as neurodegeneration. Thus, identifying new modalities to sustain NAD+ levels is critical for healthy aging.
The Cellular Consequences of Reduced NAD+
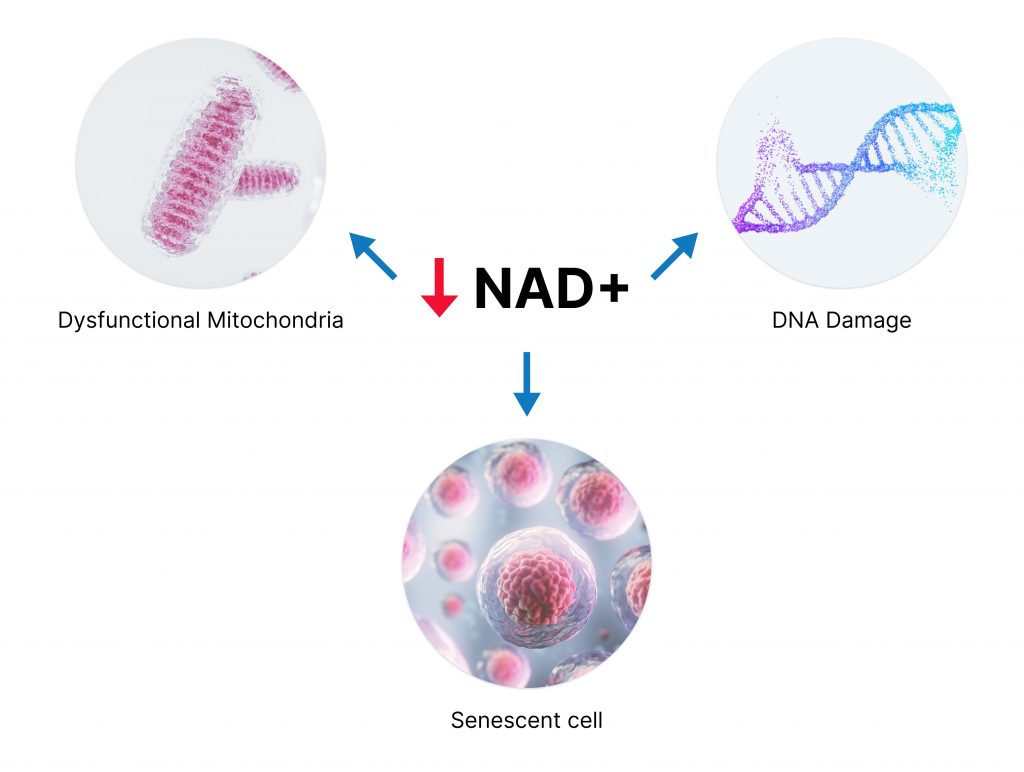
Mitochondrial Dysfunction
The removal and recycling of damaged cellular components – a process known as autophagy – is necessary to mitigate oxidative stress and inflammation, two hallmarks of aging. In particular, the autophagy of mitochondria (mitophagy) is key to ameliorating these two hallmarks and sustaining mitochondrial function, which prevents our organs from failing by supplying sufficient cellular energy (ATP). Moreover, studies have linked NAD+ depletion to mitochondrial dysfunction, which highlights the importance of replenishing the NAD+ pool to avoid the development of various age-related health conditions like sarcopenia – the progressive loss of muscle tissue.
DNA Damage and Cellular Senescence
As we age, our DNA inevitably accrues damage from a variety of factors, including solar radiation, imprecise DNA replication, and chronic inflammation. To counter DNA damage, our cells utilize the power of guardian proteins called sirtuins, which participate in activities such as cellular stress responses, circadian rhythms, and metabolism. This family of enzymes requires NAD+ for activation, thus NAD+ depletion upon aging inhibits sirtuin activity and exacerbates DNA damage.
In the absence of NAD+-powered sirtuins, chronic DNA damage leads our cells to accumulate more stress than they can handle. Eventually, the stress reaches a critical point, and damaged cells enter a state known as cellular senescence – a critical dormant state that prevents cells from growing and dividing. Studies have established senescent cell accumulation as a primary hallmark of aging, driving age-related health conditions such as neurodegeneration, diabetes, and osteoporosis – a condition that leads to brittle and weak bones.
Effects of Increased NAD+
Animal studies have shown that boosting NAD+ with precursors prolongs lifespan and improves healthspan – the number of healthy years we have. Furthermore, increased NAD+ has been shown to improve physical function, delay muscle degeneration, inhibit bone deterioration, enhance tumor suppression, and extend the lifespan of mice.
Among all age-related health conditions, humans succumb to cardiac aging the most, making it a central target in the field of aging. Two primary drivers of cardiac aging are scar-tissue buildup (fibrosis) and heart enlargement (hypertrophy). Accordingly, studies have shown that boosting NAD+ mitigates cardiac hypertrophy and delays blood vessel aging in mice.
In addition to ameliorating impaired memory, raising NAD+ levels improves cognitive function in mice modeling neurodegeneration, a deadly neurodegenerative disorder that remains to be fully understood.
Interestingly, targeting NAD+ metabolism with NAD+ boosters has proven to be effective in reducing diet-associated and age-associated weight gain in rodents. Furthermore, replenishing NAD+ levels has remedying effects on obesity in mice and reverses signs of diabetes in female mice.
Intracellular NAD+ Synthesis
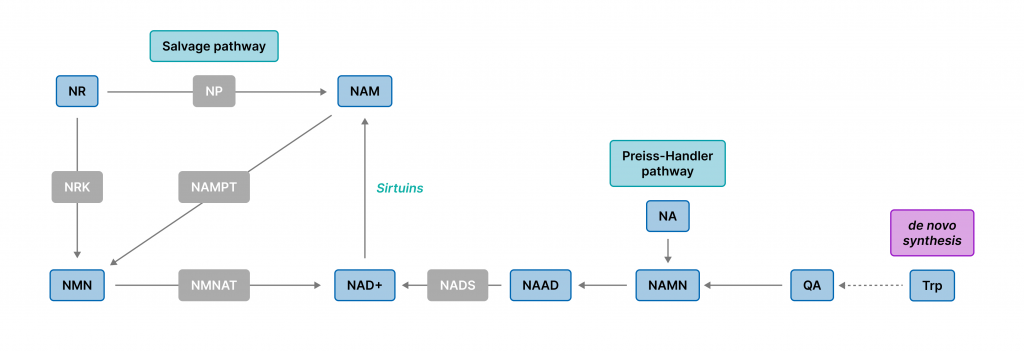
Our cells utilize precursors to synthesize NAD+ before they can occupy cellular compartments like the cytoplasm, our power generators (mitochondria), and the nucleus – the home for our DNA. NAD+ synthesis occurs via three unique pathways: the de novo pathway, the Preiss-Handler pathway, and the salvage pathway.
Amino acids (AA) are the building blocks of proteins but can also be utilized by our cells to build other molecules. The AA tryptophan can stimulate de novo NAD+ synthesis, which essentially means “from scratch.” Once obtained from dietary sources like meat, fish, and cheese, tryptophan is shuttled to the cytosol – the cell’s aqueous liquid that harbors structural components (organelles) – where it undergoes a series of chemical reactions to produce NAD+.
NAD+ synthesis can also be achieved via the Preiss-Handler pathway, where cells utilize nicotinic acid (NA), another molecule found in various foods like meat, poultry, legumes, and nuts. This pathway overlaps with the de novo pathway, as tryptophan and NA both convert to nicotinic acid mononucleotide (NAMN) before generating NAD+.
The salvage pathway is arguably the most essential NAD+ synthesis pathway, as it recycles and salvages the products of NAD+ degradation to produce more NAD+. In this setting, our cells use a family of vitamin B3’s – nicotinamide (NAM), NA, and nicotinamide ribose (NR) – to synthesize NAD+. Once NAD+ is formed, it can be broken down again to NAM, which is then converted back to NAD+’s direct precursor NMN by an enzyme called NAMPT, ultimately restarting the NAD+ loop.
Methods of NAD+ Repletion
Given NAD+’s role in multiple key metabolic pathways that contribute to energy production and DNA repair, finding ways to replenish depleting NAD+ levels upon aging is of the utmost importance.
NAD+ Precursors
One of the most potent ways to restore declining NAD+ levels is with NAD+ precursors, which have notably undergone extensive research through major institutions like Harvard. The reason for using NAD+ precursors is that NAD+ itself is not easily absorbed and cannot readily enter cells for usage. What’s more, NAD+ is quickly metabolized after oral administration, which prevents it from reaching the bloodstream. Researchers have pinpointed NMN and NR as the best NAD+ precursors, which have been shown to significantly increase circulating NAD+ in rodents and humans. Notably, studies have also shown that NMN increases physical function, reduces inflammation, and boosts cognitive function in Alzheimer’s models.
Activating NAD+ Synthesis and Inhibiting NAD+ Breakdown
Aging inevitably compromises the harmonious balance between NAD+ synthesis and NAD+ consumption, leading to reduced NAD+ levels. One of the primary drivers of NAD+ consumption upon aging is the increased activity of a surface enzyme called CD38, which breakdown NAD+ to regulate immune responses. Notably, studies have linked CD38 hyperactivation to aging pathology, prompting the use of CD38 inhibitors to not only restore NAD+ levels but also thwart aging.
Another contributor to NAD+ depletion is the increased DNA repair response that occurs upon aging. Following DNA damage, an enzyme called PARP1 attaches itself to DNA and recruits NAD+ to facilitate DNA repair. However, excess DNA damage significantly increases PARP1 activity and exacerbates declining NAD+ levels. While studies have shown that inhibiting PARP1 effectively increases NAD+, limiting PARP1 activity could slow down DNA repair, potentially accelerating the development of age-related health conditions.
One way to combat the consumption of NAD+ by enzymes like CD38 and PARP1, is to boost the activity of NAD-synthesizing enzymes. An option for achieving this is through supplementation with resveratrol, a plant-based anti-oxidant compound called a polyphenol that increases the activity of NMNAT, the enzyme responsible for converting NMN to NAD+.